Gene therapy shows promising results against multiple myeloma
For people living with relapsed or refractory multiple myeloma, a form of blood cancer that becomes increasingly difficult to treat in some people, a new investigational therapy is offering an early but encouraging sign of progress. And for eligible patients in the United States, Providence Cancer Institute is now the only site outside of Australia where they can enroll in a clinical trial evaluating it.
What makes KLN-1010 different
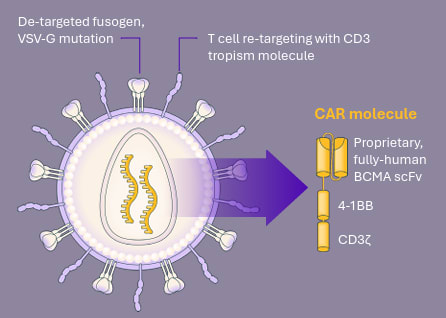
Developed by Kelonia Therapeautics, Inc., KLN-1010 is an investigational gene therapy designed to generate anti-B cell maturation antigen chimeric antigen receptor (CAR)T cells that target and destroy multiple myeloma cells.
What sets it apart is how it gets there.
Traditional CAR T therapies involve collecting a patient’s own immune cells, sending them to a lab for weeks of genetic modification, and then reinfusing them. The process can delay treatment and make access difficult for some patients.
KLN-1010 takes a different approach: it’s delivered directly by infusion, reprogramming the patient’s T cells inside the body with a single dose. Preconditioning chemotherapy isn’t required. Because it doesn't need to be custom-made for each patient, it’s considered an “off-the-shelf" therapy.
“What’s exciting about KLN-1010 is that it may simplify one of the biggest challenges with traditional CAR T therapy,” said Kevin Friedman, Ph.D., Kelonia founder and CEO. “Instead of a complex, individualized manufacturing process followed by chemotherapy, this approach is designed to work directly in the body with a single infusion. That could make this type of treatment more accessible for patients.”
Promising early results
In January 2026, the FDA-cleared-the way for KLN-1010 following a presentation of first in-human data from a phase I study at the American Society of Hematology (ASH) annual meeting. All four study participants achieved 100% minimal residual disease (MRD)-negative response rate, meaning no detectable cancer cells remained at the sensitivity levels tested.
The patients with the longest follow-up maintained those responses even at three months. A noteworthy finding came at a lower dose level, where one patient still achieved an MRD-negative response, suggesting the therapy may work effectively across a range of doses.
“While these are early results from a limited number of patients, they are encouraging,” said Dr. Friedman. “Seeing deep responses along with a manageable safety profile supports expanding this study in patients with relapsed or refractory multiple myeloma. I grew up in Portland and did my training at Providence, so it is especially meaningful to help bring this trial to patients here in Oregon.”
In terms of safety, no high-grade cytokine release syndrome or neurotoxicity was observed. Both are the more serious risks associated with traditional CAR T therapies. Low-grade side effects were manageable with standard supportive care.
Who is an eligible patient?
The phase I study underway is designed to evaluate the safety, tolerability and early effectiveness of KLN-1010 and to identify recommended doses for a future phase II study. Eligible patients must have received at least three prior lines of therapy, including a proteasome inhibitor, immunomodulatory drug, and a CD38-directed monoclonal antibody.
Enrolling now at Providence
The phase I study, known as inMMyCAR is now enrolling patients at Providence Cancer Institute. Stacy K. Lewis, M.D., is the principal investigator.
It’s worth noting that the early data offer a meaningful proof of concept for an approach that could make CAR T therapy more accessible to patients living with multiple myeloma.
Find out more about the study:
To refer a patient:-
- Call 503-215-1979
- Send an-email---